Free Energy
Free energy= Energy that can be used for anything. (Available energy)
Basic idea behind energy: Energy flows from the sun to the earth. On earth plants use the process of photosynthesis to convert the suns energy to make sugar (glucose) to build up the plants that provide energy to the heterotrophs.
[Photosynthesis->sugar-> Respiration: ATP + a little heat]
Heterotrophs:
Plants do another process called cellular respiration. It is a way to release energy found in sugars in the form of ATP. (See ATP tab)
Another important factor in the idea of free energy is maintaining a stable internal enviroment.
A stable enviroment is also known as: Homeostasis.
If you can't maintain homeostasis, you will die.
For instance our bodies keep homeostasis by sweating to cool down, or shivering when we're cold.
Plants use the "feedback method" to maintain homeostasis.
They take in their enviroment and change according to what they need.
The "Free energy equation": G=H-TS
H= Enthalpy (Total energy)
S= Entropy (Measure of disorder of a system)
Basic idea behind energy: Energy flows from the sun to the earth. On earth plants use the process of photosynthesis to convert the suns energy to make sugar (glucose) to build up the plants that provide energy to the heterotrophs.
[Photosynthesis->sugar-> Respiration: ATP + a little heat]
Heterotrophs:
Plants do another process called cellular respiration. It is a way to release energy found in sugars in the form of ATP. (See ATP tab)
Another important factor in the idea of free energy is maintaining a stable internal enviroment.
A stable enviroment is also known as: Homeostasis.
If you can't maintain homeostasis, you will die.
For instance our bodies keep homeostasis by sweating to cool down, or shivering when we're cold.
Plants use the "feedback method" to maintain homeostasis.
They take in their enviroment and change according to what they need.
The "Free energy equation": G=H-TS
H= Enthalpy (Total energy)
S= Entropy (Measure of disorder of a system)
(Less) S -> S (More energy)
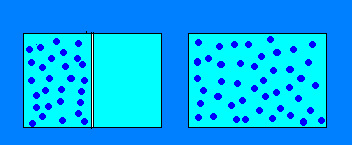
Which of these boxes has more Entropy?
Remember: Entropy= The Measure of disorder of a system
Therefore, the box without the "wall" has more Entropy.
T= Increase temperature to activate reaction.
T->T (Heat added)
Therefore, there are three variables of the free energy equation:
-Enthalpy
-Entropy
-Temperature
Which of these boxes has more Entropy?
Remember: Entropy= The Measure of disorder of a system
Therefore, the box without the "wall" has more Entropy.
T= Increase temperature to activate reaction.
T->T (Heat added)
Therefore, there are three variables of the free energy equation:
-Enthalpy
-Entropy
-Temperature