Catabolic Pathways & Oxidizing Organic Fuels
After going through the overview, this will be more in-depth about the processes.
What is the difference between fermentation and cellular respiration?
- Be careful, both of them are similar definitions.
Fermentation: Partial breaking down of sugars that occurs without oxygen
Cellular Respiration: The catabolic pathways of aerobic and anaerobic respiration, which break down organic molecules for the production of ATP.
Both of these processes break down different things, its easy to mix up these definitions.
Both cellular respiration and photosynthesis are redox reactions. In redox, reactions pay attention to the flow of electrons.
What is the difference between oxidation and reduction?
Oxidation: Loss of electrons from one substance.
Before I answer what reduction is, I will note that its pretty comical.
Its the exact opposite of what you'd assume it'd mean.
Reduction: Addition of electrons.
Typically, when you see the word "reduction" you see the word "reduce" which means smaller, or less but, in this rare case it actually means to add.
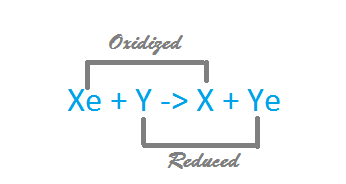
The following is a generalized formula for a redox reaction, labled with which part of the reaction is oxidized and which is reduced.
What is the difference between fermentation and cellular respiration?
- Be careful, both of them are similar definitions.
Fermentation: Partial breaking down of sugars that occurs without oxygen
Cellular Respiration: The catabolic pathways of aerobic and anaerobic respiration, which break down organic molecules for the production of ATP.
Both of these processes break down different things, its easy to mix up these definitions.
Both cellular respiration and photosynthesis are redox reactions. In redox, reactions pay attention to the flow of electrons.
What is the difference between oxidation and reduction?
Oxidation: Loss of electrons from one substance.
Before I answer what reduction is, I will note that its pretty comical.
Its the exact opposite of what you'd assume it'd mean.
Reduction: Addition of electrons.
Typically, when you see the word "reduction" you see the word "reduce" which means smaller, or less but, in this rare case it actually means to add.
The following is a generalized formula for a redox reaction, labled with which part of the reaction is oxidized and which is reduced.
Note that the electron donor is the reducing agent in this reaction, and the electron accepter is the oxidizing agent.